Amabhethri e-alkaline awaphinde ashajwe, amabhethri e-lead-asidi ayashajwa.Amabhethri e-lead-acid, aziwa nangokuthi amabhethri e-VRLA, ayahluka ngosayizi futhi ngokuvamile ane-cuboid, futhi asetshenziswa kakhulu ukuqalisa izindawo zokugcina amandla ezimotweni ezinkulu.Amabhethri e-alkaline ngokuvamile mancane futhi anosayizi oyisilinda.
Ibhethri le-asidi eholayo wuhlobo lwebhethri olunamandla kagesi aphezulu kunebhethri le-alkaline.I-voltage ephezulu ivumela ukuthi ikwazi ukunika amandla izimoto zikagesi ngamandla amaningi, futhi ikuvumela ukuthi usebenzise amandla amancane lapho usebenzisa izinto zikagesi.
Liyini Ibhethri Le-Acid Yomthofu?
Amaseli ebhethri le-asidi eholayo angase agcwale noma abe yijeli, futhi ngezinye izikhathi abizwa ngokuthi amabhethri "wet cell".I-voltage ephezulu ivumela ukuthi ikwazi ukunika amandla izimoto zikagesi ngamandla engeziwe.Amabhethri e-lead acid ayaziwa nangokuthi amaseli amanzi futhi afika ezinhlobonhlobo zamaseli ejeli.
Ibhethri ye-lead acid iwuhlobo lweibhethri elishajekayoesebenzisa amapuleti asekelwe kumthofu kanye ne-electrolyte njengomthombo wamandla.Ibhethri le-asidi eholayo linomthamo omkhulu wamandla kunezinye izinhlobo zamabhethri, okulenza libe namandla futhi lisebenze kahle.Ibhethri le-lead acid iwuhlobo lwebhethri elishajekayo elisebenzisa amapuleti omthofu njengento esebenzayo.Isetshenziswa kakhulu ezimotweni, ezikebheni nakwezinye izimoto.
Ibhethri ye-lead acid iwuhlobo lwebhethri lokugcina.Amabhethri e-lead acid athandwa kakhulu ngoba ayabiza, athembekile, futhi kulula ukuwasebenzisa.
Liyini Ibhethri Le-Alkaline?
Ibhethri le-alkaline wuhlobo lwebhethri elishajekayo elisebenzisa i-zinc chloride njenge-electrolyte yalo esikhundleni sesisombululo se-alkaline.Lokhu kwenza ibhethri le-alkaline liphephe futhi libe nobungane ngokwemvelo kunebhethri le-asidi eholayo evamile.
Ibhethri le-alkaline iseli le-electrochemical eliqukethe i-electrolyte yezinto esebenzayo equkethe usawoti wensimbi ye-alkali (i-potassium hydroxide) ne-oxide (i-potassium oxide).Angase futhi abizwe ngokuthi amabhethri eselula angashajwa noma omile ngoba awadingi ukulungiswa ngemva kokusetshenziswa.Amabhethri e-alkaline asetshenziswa kumadivayisi amaningi ahlukene, kuhlanganise namathoshi namakhamera.Sekuyiminyaka eminingi ikhona futhi izoba khona eminye eminingi.
Umehluko ekubunjweni kwebhethri:
1.Amabhethri e-lead ane-lead plate, eyenziwe ngomthofu ne-sulfuric acid.Lawa mapuleti avalelwe esitsheni esibizwa ngokuthi iseli.Uma ushaja ibhethri, i-sulfuric acid isabela namapuleti omthofu ukukhiqiza ugesi.Le nqubo yaziwa ngokuthi i-electrolysis.
2.Amabhethri e-alkaline aqukethe i-zinc ne-manganese dioxide ku-electrolyte yawo.Lezi zinto zihlangana nama-electrode (izigxobo ezinhle nezingezinhle) ukuze zikhiqize ugesi uma ushajwa kusetshenziswa ishaja.
3.Ibhethri liqukethe ama-electrode amabili kanye ne-electrolyte.I-electrode eqondile ibizwa ngokuthi i-anode, kanti i-electrode engalungile ibizwa ngokuthi i-cathode.Ebhethrini, ama-ion asuka kwenye i-electrode aye kwenye uma ufaka ugesi omncane.Lokhu kunyakaza kubizwa nge-electromotive force (EMF).
4.Ibhethri liqukethe ama-electrode amabili kanye ne-electrolyte.I-electrode eqondile ibizwa ngokuthi i-anode, kanti i-electrode engalungile ibizwa ngokuthi i-cathode.Ebhethrini, ama-ion asuka kwenye i-electrode aye kwenye uma ufaka ugesi omncane.Lokhu kunyakaza kubizwa nge-electromotive force (EMF).
5.I-voltage ekhiqizwa ibhethri iphumela kule EMF ebangela ukunyakaza phakathi kwama-electrode ayo.
Umehluko wohlelo lwebhethri:
Amabhethri e-alkaline afanele ukukhishwa okuqhubekayo nomsebenzi wamandla kagesi aphezulu, afanele amakhamera, amathoyizi kagesi, izilawuli kude, izibali, amakhibhodi, ama-shaver, njll.
Amabhethri e-lead-acid afanele izinkundla zamandla, njengamabhethri esithuthuthu, amabhethri anamandla emoto, amathoyizi kagesi emkhakheni wokugcinwa kwamandla, izinqola zegalofu zikagesi, izinhlelo ze-UPS, uchungechunge lwebhethri yethuluzi lamandla, njll.
Akushiwo ukuthi iyiphi ibhethri engcono.Uhlobo ngalunye lwebhethri lunobubanzi bohlelo lokusebenza oluhambisanayo.Iyona engcono kakhulu ukukhetha ibhethri elifanele izinkambu ezahlukene.
Impilo Yebhethri Ye-alkali:
Amabhethri e-alkaline atholakala ngamasayizi ahlukahlukene kanye nama-voltage.Anempilo yeshelufu efika eminyakeni eyi-10, uma kuqhathaniswa neminyaka emi-3 yamabhethri ajwayelekile alahlwayo.
Impilo Yebhethri Ye-Acid Eholayo:
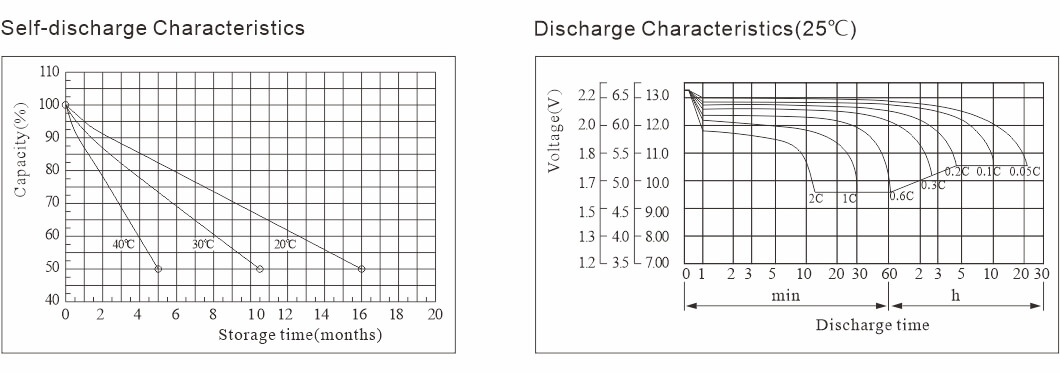
Impilo yesevisi yokuklama yamabhethri e-lead-acid yiminyaka engu-3-5 futhi ingaphezu kweminyaka engu-12, kodwa lokhu kuyimpilo yesevisi yethiyori.Kunomehluko phakathi kwempilo yesevisi yangempela kanye nethiyori.Udinga ukugcina ibhethri lakho le-asidi yomthofu kakhulu ngangokunokwenzeka ukuze uqinisekise ukuthi linokulahlekelwa okulinganiselwe okuphansi kakhulu.
Izimo zohlelo lokusebenza:
Amabhethri e-lead-acid awuhlobo oluvame kakhulu lwebhethri elisetshenziswa ezimotweni nakwezinye izinhlelo zokusebenza.Lawa mabhethri angathengwa cishe kunoma yimuphi umthengisi noma ku-inthanethi, kuye ngosayizi nohlobo olufunayo.
Ukugcinwa okuningiliziwe kwebhethri le-lead-acid kungabhekisa esihlokweni:
Uhlu Lokuhlola Lokugcinwa Kwebhethri Le-Acid Eholayo
Umehluko omkhulu phakathi kwalezi zinhlobo ezimbili zamabhethri inani lamandla agcinwe ngeyunithi ngayinye yesisindo.Ibhethri ye-lead acid ine-voltage ephezulu, okusho amandla engeziwe ukuze imoto yakho ihambe ngokushesha noma isebenzise njengesistimu yokusekela ngogesi yekhaya/ibhizinisi lakho.Amabhethri e-lead acid nawo ahlala isikhathi eside kunamabhethri e-alkaline, kodwa ngenxa yokuthi awakhiqizi amandla amaningi ngeyunithi ngayinye yesisindo, abiza kakhulu futhi!
Isikhathi sokuthumela: Jul-11-2022




